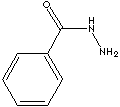
| BENZOYL HYDRAZINE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 613-94- 5 |
|
| EINECS NO. | 210-363-9 | |
| FORMULA | C6H5CONHNH2 | |
| MOL WT. | ||
| H.S. CODE | ||
|
TOXICITY |
|
|
| SYNONYMS | Benzohydrazide; Benzoic acid hydrazide; Benzhydrazide; | |
| Benzhydrazide; Benzoic acid hydrazide; | ||
| DERIVATION |
|
|
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | white to off-white crystalline powder | |
| MELTING POINT |
113 - 117 C |
|
| BOILING POINT | ||
| SPECIFIC GRAVITY | ||
| SOLUBILITY IN WATER |
|
|
| AUTOIGNITION |
|
|
| VISCOSITY | ||
| PH |
|
|
| VAPOR DENSITY | ||
| NFPA RATINGS | Health: 2; Flammability: 0; Reactivity: 0 | |
| FLASH POINT | ||
| STABILITY | Stable under ordinary conditions | |
|
GENERAL DESCRIPTION & APPLICATIONS |
||
|
Benzoic acid,
the simplest aromatic carboxylic acid containing carboxyl group bonded directly
to benzene ring, is a white, crystalline organic compound; melting at 122 C
(starting sublime at 100 C); boiling at 249 C; slightly soluble in water,
soluble in ethanol, very slightly soluble in benzene and acetone. Its aqua
solution is weakly acidic. It occurs naturally in many plants and resins.
Benzoic acid is also detected in animals. The most of commercial benzoic acid is
produced by the reaction of toluene with oxygen at temperatures around 200 C in
the liquid phase and in the presence of cobalt and manganese salts as catalysts.
It can be prepared also by the oxidation of benzene with concentrated sulphuric
acid or carbon dioxide in the presence of catalysts. Other methods are such as
by the oxidation of benzyl alcohol, benzaldehyde, cinnamic acid; by hydrolysis
of benzonitrile, benzoyl chloride. More than 90% of commercial benzoic acid is
converted directly to phenol and caprolactam. Its use in the production of
glycol benzoates for the application of plasticizer in adhesive formulations is
increasing. It is also used in the manufacture of alkyd resins and drilling mud
additive for crude oil recovery applications. It is used as a rubber
polymerization activators and retardants. Benzoic acid is converted to its salts
and esters for the use of preservative application in foods, drugs and personal
products. Sodium benzoate, sodium salt of benzoic acid, is used preferably as
one of the principal anti-microbial preservatives used in foods and beverages
(but it's concentration is limited usually not exceeding 0.1% because it is
poisonous), as it is about 200 times more soluble than benzoic acid. Sodium
Benzoate is also used in medications, anti-fermentation additives and tabletting
lubricant for pharmaceuticals. The industrial applications are as a corrosion
inhibitor, as an additive to automotive engine antifreeze coolants and in other
waterborne systems, as a nucleating agents for polyolefin, as a dye
intermediate, as a stabilizer in photographic processing and as a catalyst. Wide
range of benzoic esters are used as solvents, dying carrier, disinfectant
additive, penetrating agent and pesticides and manufacturing other compounds.
Hydrazine (anhydrous) is a clear, fuming, corrosive liquid with an ammonia-like odor; melting at 1.4 C, boiling at 113.5 C, specific gravity 1.011. It is very soluble in water and soluble in alcohol. It decomposes on heating or exposure to UV to form ammonia, hydrogen, and nitrogen, which may be explosive with a blue flame when catalysed by metal oxides and some metals such as platinum or Raney nickel. It is prepared from ammonia with chloramine in the presence of glue or gelatin (to inhibit decomposition of the hydrazine by unreacted oxidants) as the hydrate form usually (100% monohydrate contains 64% by weight hydrazine). Hydrazine is also prepared from sodium hypochlorite with urea in the presence of glue or gelatin. Botht ammonia and amines are nitrogen nucleophiles which donate electrons (they are Lewis bases). But hydrazine (diamine) has much stronger nucleophilicity which makes it more reactive than ammonia. Hydrazine has dibasic and very reactive properties. Hydrazine is used as a component in jet fuels because it produce a large amount of heat when burned. Hydrazine is used as rocket fuel. Hydrazine is used as an oxygen scavenger for water boiler feed and heating systems to prevent corrosion damage. Hydrazine is used as a reducing agent for the recovery of precious metals. It is used as a polymerization catalyst and a chain extender in urethane coatings. It and its derivatives are versatile intermediates. They have active applications in organic synthesis for agrochemicals, pharmaceuticals, photographic, heat stabilizers, polymerization catalysts, flame-retardants, blowing agents for plastics, explosives, and dyes. Recently, hydrazine is applied to LCD (liquid crystal displays) as the fuel to make faster thin-film transistors. Hydrazide is an acyl hydrazine. Acyl (-CO) is an organic radical formed by removal of a hydroxyl group from an organic acid (carboxyl group). |
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white to off-white crystalline powder | |
| ASSAY |
98.0% min |
|
| MELTING POINT |
113 - 117 C |
|
| TRANSPORTATION | ||
| PACKING | ||
| HAZARD CLASS | ||
| UN NO. | ||
| OTHER INFORMATION | ||
|
Hazard Symbols: , Risk Phrases: , Safety Phrases: 24/25 |
||